|
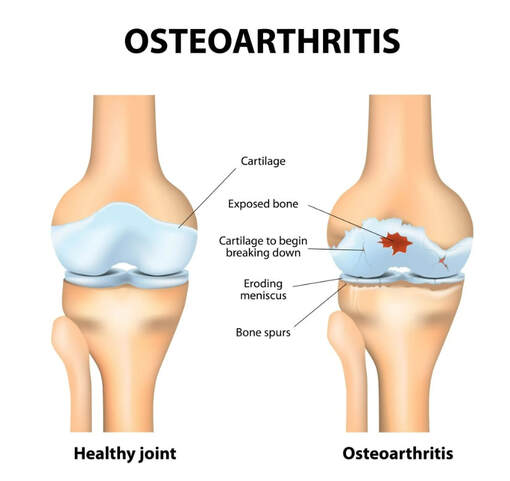
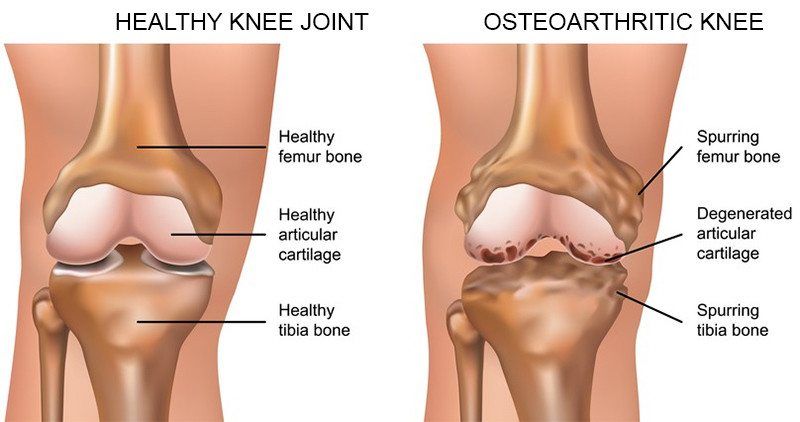
Osteoarthritis is the most common form of arthritis in the knee. It is a degenerative, "wear-and-tear" type of arthritis that occurs most often in people 50 years of age and older, although it may occur in younger people, too. In osteoarthritis, the cartilage in the knee joint gradually wears away. As the cartilage wears away, it becomes frayed and rough, and the protective space between the bones decreases. This can result in bone rubbing on bone, and produce painful bone spurs. Osteoarthritis usually develops slowly and the pain it causes worsens over time. I know it can be hard to determine if your patient's injection would need a prior authorization or precertificaton. It is always best that we verify our patients' coverage, benefits and eligibility. PRIOR AUTHORIZATION, PRECERTIFICATION & BILLING PROCESS - HYALURONAN ACID & VISCO THERAPIES FOR OSTEOARTHRITIS OF THE KNEE CPT CODES: 20605, 20606, 20610, 20611 HCPCS CODES: J7318, J7323, J7328, J3490, J7320, J7321, J7322, J7324, J7325, J7326, J7327, J7329, J7331, J7332 Let's look at a commonly published clinical policy and guideline for these visco and hyaluronan acid therapies. Pay attention to everything on this post because these can all be very helpful for your billing and in order to maximize reimbursement. Conservative Treatment: Physical therapy or pharmacotherapy/NSAIDs (e.g., non-steroidal anti-inflammatory drugs [NSAIDs], acetaminophen and/or topical capsaicin cream) Understanding Indications and Dosing: Intra-articular injections of sodium hyaluronate are proven and medically necessary when all of the following are met: 1. Diagnosis of knee osteoarthritis 2. Member has not responded adequately to conservative therapy which may include physical therapy or pharmacotherapy or injection of intra-articular steroids or member is unable to tolerate conservative therapy because of adverse side effects 3. The member reports pain which interferes with functional activities. --- ambulation, prolonged standing 4. The pain is attributed to degenerative joint disease/primary osteoarthritis of the knee. 5. There are no contraindications to the injections --- active joint infection, bleeding disorder 6. Dosing is in accordance with the US FDA approved labeling as follows. --- Durolane: Approved as a single injection --- Euflexxa: Approved for 3 injections --- Gel-One: Approved as a single injection --- Gelsyn-3: Approved for 3 injections --- GenVisc 850: Approved for 3-5 injections --- Hyalgan: Approved for 5 injections --- Hymovis: Approved for 2 injections --- Monovisc: Approved as a single injection --- Orthovisc: Approved for 3-4 injections --- Supartz: Approved for 3-5 injections --- Synojoynt: Approved for 3 injections --- Synvisc One: Approved as a single injection --- Synvisc: Approved for 3 injections --- Triluron: Approved for 3 injections --- TriVisc: Approved for 3 injections --- Visco-3: Approved for 3 injections Learn how to properly bill and report these HCPCS codes for maximized reimbursement. READ HERE! Common Criteria and Utilization Limitations: Initial Determination: --- therapy has not resulted in functional improvement after at least 3 months Reauthorization/Continuation --- At least 6 months have passed since the prior course of treatment for the respective joint Intra-articular injections of sodium hyaluronate are unproven and not medically necessary for treating any other indication due to insufficient evidence of efficacy including, but not limited to the following: ----- Hip osteoarthritis ----- Temporomandibular joint osteoarthritis ----- Temporomandibular joint disc displacement Hyaluronic acid gel preparations to improve the skin's appearance, contour and/or reduce depressions due to acne, scars, injury or wrinkles are considered cosmetic and are not covered. Contraindications: 1. Do not administer to patients with known hypersensitivity (allergy) to hyaluronate preparations or allergies to avian or avian-derived products (including eggs, feathers, or poultry). This contraindication does not apply to Orthovisc. 1. Do not administer to patients with known hypersensitivity (allergy) to gram positive bacterial proteins. This contraindication applies to Orthovisc only. 1. Do not inject sodium hyaluronate into the knees of patients with infections or skin diseases in the area of the injection site or joint. Imaging Requirements: (MRI, CT, XRAY, U/S):X-ray, CT, or MRI reports), Color Doppler ultrasound (CDUS) and synovitis scores Pain Relief: Knee Osteoarthritis (OA) --- single 6 ml IA injection of hylan G-F 20 provided better pain relief over 26 weeks Sodium Hyaluronate Product Therapy Using Preferred Products (Durolane, Euflexxa, and Gelsyn-3) >>Medical notes documenting all of the following: --- Current prescription --- diagnosis of OA of the knee --- Conservative treatment tried for at least 3 months including response --- Signs and symptoms --- Current functional limitations --- Complete report(s) of diagnostic imaging (X-ray, CT, or MRI reports) --- Previous sodium hyaluronate treatment provided including the brand name of the drug, course of treatment, and response --- Dose, frequency, interval since previous sodium hyaluronate treatment --- Physician treatment plan Medical Necessity Cross-over: M17.0 -- Bilateral primary osteoarthritis of knee M17.10 -- Unilateral primary osteoarthritis, unspecified knee M17.11 -- Unilateral primary osteoarthritis, right knee M17.12 -- Unilateral primary osteoarthritis, left knee M17.2 -- Bilateral post-traumatic osteoarthritis of knee M17.30 -- Unilateral post-traumatic osteoarthritis, unspecified knee M17.31 -- Unilateral post-traumatic osteoarthritis, right knee M17.32 -- Unilateral post-traumatic osteoarthritis, left knee M17.4 -- Other bilateral secondary osteoarthritis of knee M17.5 -- Other unilateral secondary osteoarthritis of knee M17.9 -- Osteoarthritis of knee, unspecified Learn how to Bill for HYALURONAN ACID & VISCO THERAPIES FOR OSTEOARTHRITIS OF THE KNEE References: Clinical Policies of Commercial Payers and Medicare from published in the public domains. CPT Codes are a trademark and owned by the American Medical Association. ICD-10 Code book of 2022. Categories All Archives July 2024
0 Comments
Your comment will be posted after it is approved.
Leave a Reply. |

ABOUT THE AUTHOR:
Ms. Pinky Maniri-Pescasio is the Founder of GoHealthcare Consulting. She is a National Speaker on Practice Reimbursement and a Physician Advocate. She has served the Medical Practice Industry for more than 25 years as a Professional Medical Practice Consultant. search hereArchives
July 2024
Categories
All
BROWSE HERE
All
|
- About
- Leadership
- Contact Us
- Testimonials
- READ OUR BLOG
-
Let's Meet in Person
- 2023 ORTHOPEDIC VALUE BASED CARE CONFERENCE
- 2023 AAOS Annual Meeting of the American Academy of Orthopaedic Surgeons
- 2023 ASIPP 25th Annual Meeting of the American Society of Interventional Pain Management
- 2023 Becker's 20th Annual Spine, Orthopedic & Pain Management-Driven ASC Conference
- 2023 FSIPP Annual Conference by FSIPP FSPMR Florida Society Of Interventional Pain Physicians
- 2023 New York and New Jersey Pain Medicine Symposium
- Frequently Asked Questions and Answers - GoHealthcare Practice Solutions
- Readers Questions
Photos from shixart1985 (CC BY 2.0), www.ilmicrofono.it, shixart1985




 RSS Feed
RSS Feed 